Ontario leads race to develop stem cell therapy for spinal cord injuries
By Lisa Willemse
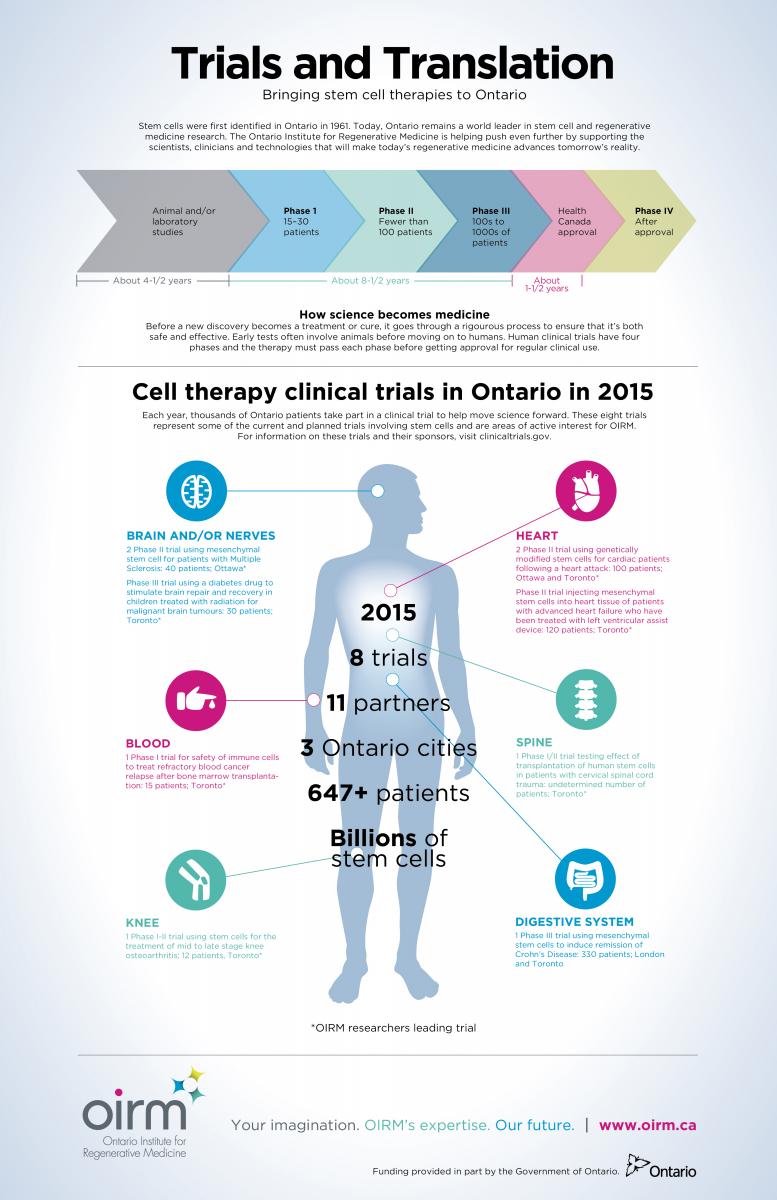
As recently as 10 years ago, the announcement of a stem cell clinical trial for a condition other than cancer would have been front-page news across the globe. While many such announcements are still newsworthy, the steady increase in new stem cell trials for a range of conditions has made them a much more common occurrence. Consider that this year, in Ontario alone, there are at least eight such studies underway or scheduled to begin.

This surge can be attributed to a few key factors. First is the deep strength of stem cell and regenerative medicine research in Ontario, which ranks exceptionally high internationally. World-class clinical infrastructure and a supportive regulatory environment, also play an important role. Finally, there is a considerable coordinated focus on clinical and commercial translation on the part of government, research and health organizations, as well as business and innovation enterprises. All of this makes Ontario an attractive place to conduct stem cell clinical trials.
“It’s a significant reflection on Ontario’s vision for the future that the province recognizes the potential of regenerative medicine and is investing in the creation of a biotech environment that supports clinical translation and commercialization of research,” says Dr. Michael Fehlings, a clinician-researcher at the University Health Network in Toronto. Fehlings is the lead researcher in an ongoing clinical trial, sponsored by Stem Cells Inc., which is testing the safety of neural stem cells used to regenerate thoracic and cervical spinal cord injuries.
The first phase of the trial, now concluded and being written for publication, enrolled 12 participants with thoracic spinal cord injuries in Toronto, Calgary and Zurich. The second phase is slated to start later this year in Toronto and other yet-to-be-identified centres, and will enrol participants with a cervical spinal cord injury. Dr. Fehlings and his team are excited to launch the second phase for several reasons, and paramount among these is the potential impact: injuries to the cervical spine account for approximately 70% of all spinal cord injuries in Canada. Such injuries often result in quadriplegia, with significant reduction in quality of life for those afflicted.
“I think this could have a big impact because the cervical spinal cord is so eloquent and even small amounts of regeneration — of even one or two centimetres — can influence one or two segments of the spinal cord. So, if this therapy, for example, can achieve an improvement from a C6- to a C8-level injury, they will regain the use of their hands, which can have a huge impact on overall recovery,” says Dr. Fehlings.
“To put this in context with the first study, an equivalent improvement on the thoracic spinal cord might not be detectable at all by the patient or the physician. So the impact here for the cervical spinal cord is enormous.”
The trial is one of several with connections to the Ontario Institute for Regenerative Medicine, which was formed in 2014 to coordinate Ontario’s research, clinical and commercial efforts in making Ontario a global leader in the development of stem cell-based products and therapies. OIRM has focused its energies in advancing stem cell and regenerative medicine therapies for a range of debilitating diseases for which current treatments fall short. These include autoimmune disorders, and diseases or injuries that affect musculoskeletal, cardiovascular, vision, neurological and blood systems. Within the current list of stem cell clinical trials, OIRM researchers are playing lead roles in all but one [see infographic].
This commitment will bring a wealth of new knowledge that in turn can be applied to future studies.
It may well mean that for people suffering with ailments such as heart disease, Crohn’s, Multiple Sclerosis — and significantly for those with cervical spinal cord injuries — a treatment will one day be within their grasp.
*This article may be reprinted in full or in part with credit to the author and to the Ontario Institute for Regenerative Medicine. Please include link to original source.
*Click on infographic picture to download it as a pdf file.
Update June 15, 2016. Additional trials have developed in Ontario. To see the updated list click here.