Cells, cell soup and treating lung inflammation
Read our latest article on lung repair using stem cells.
By Meredith Hanel
In regenerative medicine choosing the right cell for the job depends on the right fit between the disease type and the qualities of the cell. Mesenchymal stromal cells (sometimes called mesenchymal stem cells or MSCs) have turned out to be a great fit for several lung diseases. Strangely, the job they do best — calming down inflammation — is not what they were initially selected to do.
MSCs are found in many adult tissues, including bone marrow and umbilical cords, and have the ability to create a variety of cell types, which is why scientists were very excited about them initially. But when OIRM researcher and neonatologist Dr. Bernard Thébaud at The Ottawa Hospital began testing MSCs in animal models for lung disease, the cells didn’t perform as expected. “We were surprised because we thought the cells would go into the lungs and replace the dead cells and become new lung cells but then when we labelled them with fluorescent dye and looked at the lungs we couldn’t find those cells anymore.” The MSCs didn’t stick around for more than a few days, yet they were clearly having an effect.
 |
| Watch: Short animated video featuring Marissa Lithopoulos, PhD candidate in Dr. Bernard Thébaud’s lab, explaining the use of stem cells to treat bronchopulmonary dysplasia |
Meanwhile evidence was building about different molecules that MSCs might secrete into their surroundings including those factors with anti-inflammatory properties. Other research showed MSCs can migrate to the sites of injury, communicate with surrounding cells and respond accordingly. So MSCs aren’t passively sitting there leaking molecules into their environment, they are more like medical devices delivering appropriate medicinal factors when needed.
Dr. Thébaud researches umbilical cord derived MSCs for treatment of bronchopulmonary dysplasia (BPD) also known as chronic lung disease of prematurity, the most frequent complication of extreme prematurity in infants.
The lungs of these extremely premature infants, born at less than 28 weeks, are extremely fragile and great care must be taken to avoid injury and infections, including possible side-effects of a therapeutic intervention, such as MSCs.
Thébaud and his team wondered if the culture medium that MSCs were grown in would be beneficial even without the cells, akin to how water added to soup becomes a flavourful and nutritious broth. “We took the cell-free soup and injected this soup into our animal model and we showed the same therapeutic benefit that we had with the cells,” explains Thébaud.
“We thought, if we give only the soup it might be a safer approach because we don’t provide any cells that could do some harm maybe many years down the road. On the other hand, we might not capitalize fully on the therapeutic potential of the cells.”
The potential of MSCs to cause harm is an ongoing question. An early fear that they might cause tumours is beginning to disappear now that more than a thousand people have been treated with MSCs. But Thébaud, who also holds positions at the Children’s Hospital of Eastern Ontario (CHEO) and the University of Ottawa, is especially cautious, saying that, “Thinking about treating babies, it always stays on my mind,” and further explaining that the behaviour of transplanted cells in the environment of a very young baby may be different than in an adult. “That’s why we have to have very carefully designed trials and place all babies into a registry so we can follow them for their whole life,” Thébaud says.
“To keep these babies alive we have to mechanically ventilate them and give them oxygen and while this saves their lives it also can damage their lungs.”
Infections further impede lung growth contributing to this inflammatory disease. MSCs, in addition to the calming inflammation, produce factors which can promote lung growth. So far a small phase I clinical trial in Korea suggests treating BPD with umbilical cord MSCs is feasible and safe.
To convince himself and regulatory agencies that umbilical MSCs are safe for babies, Thébaud is first testing MSCs in primates delivered prematurely, intubated and mechanically ventilated exactly how human babies are treated in the neonatal intensive care unit. Thébaud’s team is testing the best way to deliver the cells and prevent the disease in these animals and apply that knowledge to human infants. If all continues to go well, Thébaud is planning a BPD clinical trial in humans in 2018.
The circulation subway station: MSCs stop at the lungs
Over in Toronto, Dr. John Laffey, anesthesiologist, critical care physician and OIRM researcher at St. Michael’s Hospital, is developing umbilical cord MSCs to treat acute respiratory distress syndrome (ARDS). Right now clinical trials for sepsis and ARDS use bone-marrow-derived MSCs. They are the most well studied but getting bone marrow cells is an invasive procedure. Umbilical cord MSCs work as well as bone marrow derived MSCs in Laffey’s animal models.
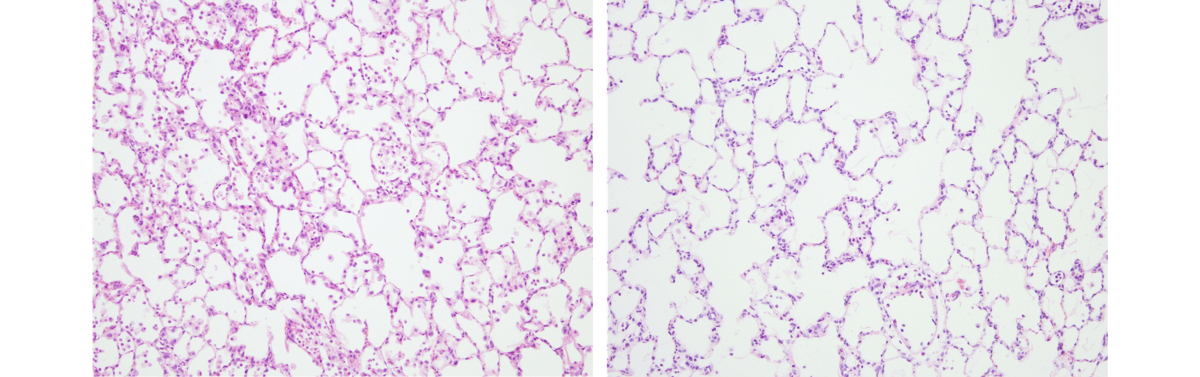 |
| Thin slices through rat lungs. Injured rat lungs (left) are filled with more cells because of blood and pus associated with inflammation but lungs repaired by mesenchymal stromal cells (right) have more clear open spaces. Courtesy of Dr. John Laffey. |
A person can be given their own MSCs (autologous) or from an unrelated recipient (allogeneic) without immunosuppressive drugs. “That’s very important in acute diseases like ARDS or sepsis where the patient really does not have the time needed to prepare autologous therapies.” Since pneumonia and sepsis are major causes of ARDS, other ongoing work in those areas, such as a clinical trial testing bone marrow derived MSCs for sepsis taking place in Ottawa should produce findings that will be very relevant to ARDS.Laffey and his team of researchers are applying for OIRM funding for clinical testing of umbilical cord derived MSCs in ARDS. Laffey says the collection of tertiary critical care facilities in close proximity in Toronto is “a huge advantage in terms of being able to do an early phase study literally within the city boundaries.”
Rather than putting MSCs directly into the lungs, Laffey’s team plans to administer umbilical cord MSCs intravenously. It may seem like a roundabout way of getting to the lungs but it works just as well and it may be better tolerated by the patient than pushing a large volume of fluid containing millions of cells into the lung. Circulation is like a subway system that passes through the lungs. Conveniently for lung researchers, most MSCs get off at “lung station” where they become trapped because of their size and stickiness, but not so convenient for other researchers, such as cardiologists who want to deliver MSCs to the heart.
In animal studies MCSs remain in the lung for about 48 hours and for acute diseases this is long enough. For chronic diseases, like chronic obstructive pulmonary disease, lung fibrosis and asthma, it may be necessary to give repeat injections but no one knows yet. Even if they don’t stick around MSCs seem to have long term benefits and researchers think they might protect or repair the resident stem cells, making them healthy enough to further contribute to lung regeneration.
Many lung diseases involve inflammation and dealing with inflammation is what MSCs do best. This is why MSCs hold promise for so many diseases outside the lung as well. Researchers are still trying to work out all their potential and their limitations.
“It is through these clinical trials and the return to the bench to study the mechanism of action of these cells that we will learn exactly how to best use MSCs”, says Thébaud. Clinical testing of MSCs in lung disease is only at the beginning. If proven safe and effective the next hurdle will be scaling up to actually treat the large numbers of patients that will need them.
