Ontario-Made Innovation Takes on COVID-19
Cell therapy for severe infection only possible with significant investments in stem cell research
 Like far too many Canadians, COVID-19 has been an arduous road for Ottawa native Sharon Charlebois. More than 10 months after her 25-day admission to The Ottawa Hospital in January 2021 – which included a trip to the intensive care unit (ICU) – she struggles with significant, lingering effects.
Like far too many Canadians, COVID-19 has been an arduous road for Ottawa native Sharon Charlebois. More than 10 months after her 25-day admission to The Ottawa Hospital in January 2021 – which included a trip to the intensive care unit (ICU) – she struggles with significant, lingering effects.
“I’m definitely what they call a long-hauler,” she says. “It really is not a pleasant disease. When I came home from the hospital, I needed a personal support worker every day, and physiotherapy. Today, I deal with post-traumatic stress disorder and memory loss – I’m searching for the right words all the time. I’m breathless, too, and use a walker when I am outside. It takes me twice as long to do something. I’m certainly not myself.”
While Sharon works on her recovery, she notes that she is happy to be alive – and credits a cell therapy developed by researchers at The Ottawa Hospital she received as part of a phase-1 randomized clinical trial now underway in Ottawa, Montreal and Toronto.
The trial, led by The Ottawa Hospital’s Executive Vice-President of Research Dr. Duncan Stewart, is designed to assess the effectiveness of using mesenchymal stem/stromal cells (MSCs) to treat severe lung inflammation in COVID-19 patients. The treatment was originally developed to treat runaway inflammation in patients with sepsis, which can cause Acute Respiratory Distress Syndrome (ARDS) and subsequent respiratory and organ failure. ARDS also affects 80% of COVID-19 patients in the ICU and nearly half of those with COVID-19 related pneumonias. It is often fatal. Fortunately, the Ontario Institute for Regenerative Medicine (OIRM) invested $2.2M over five years into the development of this treatment for sepsis, which allowed researchers to quickly pivot the therapy to treat patients with severe COVID-19.
When Sharon was admitted to the hospital’s COVID-19 ward, she was started on oxygen right away. On the second day, she collapsed while attempting to stand and was immediately brought to the ICU. Her family was told the next 24 hours would be critical. Her family was approached by a staff nurse and ICU research coordinator to participate in stem cell research, including for the MSC trial. Sharon’s husband then spoke to her niece, also a nurse, who encouraged Sharon to participate. On her fourth day in hospital, she received the first of three MSC infusions to be given once daily over the next three days. On the fifth day, she also had a feeding tube inserted, and became the first patient at the hospital to be given a ventilation helmet set at 100% oxygen to assist with breathing.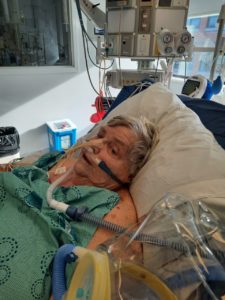
“I don’t remember much about my time in the hospital – but bits and pieces come back to me,” Sharon says. “They seemed to think the stem cells might help, and I thought, even if it doesn’t work on me, it might help somebody else.”
Sharon says she responded within days after the infusions – an oxygen helmet she was wearing was removed the day after the third infusion.
Over the next several days, she required less and less supplemental oxygen to breathe. By the 16th day, Sharon was out of bed for the first time since being admitted to the ICU. By the 18th day, she was out of the ICU.
While cautioning that results of the clinical trial are not expected until some time in 2022, Dr. Duncan Stewart notes that Sharon’s story would not be possible without strong support for stem cell research in Ontario from the provincial government.
“Solving complex scientific challenges to create new treatments that save lives requires a significant, sustained investment, particularly if we want to see Ontario-made technologies stay in Ontario for the benefit of our health and the economy,” Sandra Donaldson, Vice President & Chief Operating Officer says. “We have world-class research and cell manufacturing in Ontario, as well as cutting-edge research hospitals and patients like Sharon who are willing to participate in trials – but we need that sustained commitment from the top to reach our true potential.”
Sharon adds that she is grateful for the care and attention she received on the COVID-19 ward, in the ICU and from the clinical trial team.
“I cannot believe the level of care I received – they made me so comfortable. And I am convinced the stem cells helped me, because I was feeling so much better quickly, and I remember thinking it has to be because of the infusions,” she concludes. “I believe the treatment worked, and so does my family.”
