2019 – 2021 Funded Post-Doctoral Fellowship Projects
Deconstructing the myogenic lineages at single cell resolution
Researcher: Alex Lin
Institute: Ottawa Hospital Research Institute
Supervisor: Michael Rudnicki
Award Value: $150,000
 Adult skeletal muscle has the remarkable capacity to regenerate in response to injury, which is imparted by muscle stem cells (MuSCs). Even though adult skeletal muscle is a diverse tissue comprised of different muscle groups with various fiber type compositions, our understanding of how a MuSC makes a myofiber is a linear process with sequential activation of transcription factors. This prevailing dogma of linear activation during MuSC commitment cannot account for the functional diversity observed at the tissue level or the bipotential activity of MuSCs, such as making brown fat. We hypothesize that MuSCs are primed and exist in discrete subpopulations that differentiate down multiple lineages. The existence of subpopulations suggests population heterogeneity of the MuSCs and a hierarchical organization with distinct functional properties. To address this our work will combine classical lineage tracing experiments with molecular profiling and characterization. Use of single cell RNA sequencing will help to unravelling the complexities by profiling single cells instead of heterogeneous bulk populations. We hypothesize that using scRNAseq of MuSCs in combination with lineage tracing models; will uncover novel subpopulations and their regulators, and ultimately, deconstruct the myogenic lineages.
Adult skeletal muscle has the remarkable capacity to regenerate in response to injury, which is imparted by muscle stem cells (MuSCs). Even though adult skeletal muscle is a diverse tissue comprised of different muscle groups with various fiber type compositions, our understanding of how a MuSC makes a myofiber is a linear process with sequential activation of transcription factors. This prevailing dogma of linear activation during MuSC commitment cannot account for the functional diversity observed at the tissue level or the bipotential activity of MuSCs, such as making brown fat. We hypothesize that MuSCs are primed and exist in discrete subpopulations that differentiate down multiple lineages. The existence of subpopulations suggests population heterogeneity of the MuSCs and a hierarchical organization with distinct functional properties. To address this our work will combine classical lineage tracing experiments with molecular profiling and characterization. Use of single cell RNA sequencing will help to unravelling the complexities by profiling single cells instead of heterogeneous bulk populations. We hypothesize that using scRNAseq of MuSCs in combination with lineage tracing models; will uncover novel subpopulations and their regulators, and ultimately, deconstruct the myogenic lineages.
2016 – 2019 Fellowships
Local ON/OFF control of tolerance toward allograft cell transplants
Jeffrey Harding, Lunenfeld-Tannenbaum Research Institute
Supervisor: Andras Nagy
Dissecting the role of Notch signaling during the transition from HE to HSC
Vladimir Manchev, University Health Network
Supervisor: Gordon Keller
2017-2019 (co-funded by Medicine by Design):
Rescuing vision in models of retinal degeneration
Project Leader: Arturo Ortin-Martinez, University Health Network (Valerie Wallace lab)
Developing an immune-cell approach to cardiac regeneration
Project Leader: Yiming Wang, University Health Network (Slava Epelman lab)
2018 – 2020 Fellow
Discovering novel regulators of human totipotent stem cells
Project Leader: Tatsuya Yamakawa, The Hospital for Sick Children (Janet Rossant)
Other Current Funding:
Disease Teams
New Ideas Grants
2019-2020 New Ideas Project Awardees
Discovering genetic factors that regulate axon regeneration in stem cell derived human neurons
Investigator: Yun Li
Institute: Hospital for Sick Children
Award Value: $75,000
 Damaged neurons in the adult human central nervous system (CNS) do not readily regenerate. This lack of regeneration limits the functional recovery for CNS injuries such as stroke, traumatic brain and spinal cord injuries, multiple sclerosis, infection and other related conditions that involve axon pathology. The lack of ability for human neurons to regrow their axons is a major therapeutic limitation. However, recent animal studies suggest that lack of axon regeneration is the result of intrinsic inhibitory signals (blocking signals) that can be neutralized. These findings raise the exciting prospect that human CNS neurons can be coaxed into regenerating their axons if the inhibitory blockades can be identified and removed. In this project, a genome-wide search for all intrinsic inhibitory factors that prevent axon regeneration will be conducted. This will be accomplished by establishing an in vitro axon injury and regeneration assay using CNS neurons derived from human pluripotent stem cells and will be subjected to a genome-wide CRISPR knockout screen. This project seeks to identify genes which when deleted, allow the human axons to regenerate. This research will establish a genome-wide approach to study the genetic regulation of human axon regeneration, and identify targets for potential therapeutic intervention for a wide spectrum of CNS diseases that involve axon pathology.
Damaged neurons in the adult human central nervous system (CNS) do not readily regenerate. This lack of regeneration limits the functional recovery for CNS injuries such as stroke, traumatic brain and spinal cord injuries, multiple sclerosis, infection and other related conditions that involve axon pathology. The lack of ability for human neurons to regrow their axons is a major therapeutic limitation. However, recent animal studies suggest that lack of axon regeneration is the result of intrinsic inhibitory signals (blocking signals) that can be neutralized. These findings raise the exciting prospect that human CNS neurons can be coaxed into regenerating their axons if the inhibitory blockades can be identified and removed. In this project, a genome-wide search for all intrinsic inhibitory factors that prevent axon regeneration will be conducted. This will be accomplished by establishing an in vitro axon injury and regeneration assay using CNS neurons derived from human pluripotent stem cells and will be subjected to a genome-wide CRISPR knockout screen. This project seeks to identify genes which when deleted, allow the human axons to regenerate. This research will establish a genome-wide approach to study the genetic regulation of human axon regeneration, and identify targets for potential therapeutic intervention for a wide spectrum of CNS diseases that involve axon pathology.
Progressive supranuclear palsy drug target testing using human neurons
Investigator: Yu Lu
Institute: McMaster University
Co-investigators: Karun Singh, McMaster University
Award Value: $75,000
 Progressive supranuclear palsy (PSP) is a neurodegenerative disease affecting mobility, behavior, speech, and thinking. The brain of a PSP patient contains many dead or dying brain cells with internal protein aggregates. The main component of these aggregates is a protein called Tau, which, in a healthy brain, supports vital cell transport systems inside the brain cells. However, Tau aggregates in patients with PSP cause brain cell dysfunction and death. Currently, there is no medication for the core symptoms of PSP.
Progressive supranuclear palsy (PSP) is a neurodegenerative disease affecting mobility, behavior, speech, and thinking. The brain of a PSP patient contains many dead or dying brain cells with internal protein aggregates. The main component of these aggregates is a protein called Tau, which, in a healthy brain, supports vital cell transport systems inside the brain cells. However, Tau aggregates in patients with PSP cause brain cell dysfunction and death. Currently, there is no medication for the core symptoms of PSP.
In our brain cells, the Tau gene encoded in the DNA is first used to make message molecules known as RNA. These Tau RNA molecules go through a process called splicing, during which these RNA molecules are edited. Multiple proteins, many still unknown, control splicing of the Tau RNA molecule. Any mistakes in the levels and functions of these proteins could lead to out-of-control Tau splicing, which is a major cause of Tau aggregate formation and brain dysfunction. Therefore, it is important to develop drugs to tackle the aberrant Tau splicing in PSP. In this study, the objective is to use a human stem cell model to unveil proteins contributing to Tau splicing mistakes in PSP patients. These proteins could represent potential new drug targets to correct Tau splicing and treat PSP.
Treating aberrant brain development associated with congenital muscle disease by sensorimotor stimulation
Investigator: Vince Tropepe
Institute: University of Toronto
Award Value: $73,000
 Congenital myopathies and muscular dystrophies (CMDs) are heterogeneous disorders, many of which have neurodevelopmental co-morbidities. Defects in brain development, such as cerebral hypoplasia, are often considered secondary to the primary muscle disease phenotype. Previous research has discovered that movement is critical for promoting normal brain development (neurogenesis) and growth of the post-embryonic forebrain. Restricting movement caused premature differentiation of neural progenitors and brain dysfunction. Movement-dependent forebrain neurogenesis requires somatosensory feedback from the body. Such a relationship between movement and brain development might explain the known co-morbidity between physical and neurological deficits in congenital muscle diseases (CMDs). We propose to characterize the post-embryonic forebrain growth in established models of CMD and test the possibility of combatting aberrant neurodevelopment by providing sensorimotor input to the brain. To do so, we will characterize the changes underlying neurogenesis leading to a reduction in forebrain growth in selected CMD mutants. Second, we will test if aberrant brain development associated with CMD can be combatted via targeted activation of dorsal root ganglion (DRG) neurons and finally, we will examine whether norepinephrine is a key mediator of sensorimotor dependent forebrain neurogenesis. This research will reveal the neurodevelopmental consequences of CMD and provide novel therapeutic avenues for promoting healthy brain growth in CMD patients.
Congenital myopathies and muscular dystrophies (CMDs) are heterogeneous disorders, many of which have neurodevelopmental co-morbidities. Defects in brain development, such as cerebral hypoplasia, are often considered secondary to the primary muscle disease phenotype. Previous research has discovered that movement is critical for promoting normal brain development (neurogenesis) and growth of the post-embryonic forebrain. Restricting movement caused premature differentiation of neural progenitors and brain dysfunction. Movement-dependent forebrain neurogenesis requires somatosensory feedback from the body. Such a relationship between movement and brain development might explain the known co-morbidity between physical and neurological deficits in congenital muscle diseases (CMDs). We propose to characterize the post-embryonic forebrain growth in established models of CMD and test the possibility of combatting aberrant neurodevelopment by providing sensorimotor input to the brain. To do so, we will characterize the changes underlying neurogenesis leading to a reduction in forebrain growth in selected CMD mutants. Second, we will test if aberrant brain development associated with CMD can be combatted via targeted activation of dorsal root ganglion (DRG) neurons and finally, we will examine whether norepinephrine is a key mediator of sensorimotor dependent forebrain neurogenesis. This research will reveal the neurodevelopmental consequences of CMD and provide novel therapeutic avenues for promoting healthy brain growth in CMD patients.
Generation of heart pacemaker cells from stem cells
Investigator: Stephanie Protze
Institute: University Health Network
Award Value: $75,000
 The human heart rate is controlled by two specialized pacemaker tissues in the heart known as the sinoatrial node (SAN) and the atrioventricular node (AVN). The SAN initiates electrical signals that drive the heartbeat while the AVN is responsible for propagating these electric signals from the upper heart chambers to the bottom heart chambers. This activates their contraction and results in the pumping of blood through the body. Failure of either pacemaker tissue leads to a life-threatening irregular or slow heartbeat and needs to be treated by implantation of an electronic pacemaker device. Approximately 21,000 Canadians receive an electronic pacemaker every year. While representing an effective treatment, electronic pacemakers have a number of disadvantages including the risk of pacemaker lead infections, the need for surgical battery replacements, and the limited adaption to growth in cases of pediatric patients. The ability to replace failing pacemaker tissue with functional pacemaker cells derived from stem cells to generate a biological pacemaker represents an attractive alternative that could overcome these disadvantages. We have previously been successful in generating SAN pacemaker cells from stem cells and have shown that these cells can function as a biological pacemaker in a small animal model. In this study we aim to identify methods to derive AVN pacemaker cells from stem cells and to functionally characterize their pacemaker phenotype. The long-term objective is to use these AVN pacemaker cells in biological pacemaker approaches to provide an improved treatment for patients with AVN pacemaker diseases in Canada and worldwide.
The human heart rate is controlled by two specialized pacemaker tissues in the heart known as the sinoatrial node (SAN) and the atrioventricular node (AVN). The SAN initiates electrical signals that drive the heartbeat while the AVN is responsible for propagating these electric signals from the upper heart chambers to the bottom heart chambers. This activates their contraction and results in the pumping of blood through the body. Failure of either pacemaker tissue leads to a life-threatening irregular or slow heartbeat and needs to be treated by implantation of an electronic pacemaker device. Approximately 21,000 Canadians receive an electronic pacemaker every year. While representing an effective treatment, electronic pacemakers have a number of disadvantages including the risk of pacemaker lead infections, the need for surgical battery replacements, and the limited adaption to growth in cases of pediatric patients. The ability to replace failing pacemaker tissue with functional pacemaker cells derived from stem cells to generate a biological pacemaker represents an attractive alternative that could overcome these disadvantages. We have previously been successful in generating SAN pacemaker cells from stem cells and have shown that these cells can function as a biological pacemaker in a small animal model. In this study we aim to identify methods to derive AVN pacemaker cells from stem cells and to functionally characterize their pacemaker phenotype. The long-term objective is to use these AVN pacemaker cells in biological pacemaker approaches to provide an improved treatment for patients with AVN pacemaker diseases in Canada and worldwide.
Development of decellularized scaffolds for intervertebral disc repair and regeneration
Investigator: Lauren Flynn
Institute: Western University
Co-investigators: Cheryle Séguin, Western University
Award Value: $75,000
 Chronic back pain associated with intervertebral disc degeneration is a debilitating condition that negatively impacts the quality of life of many Canadians. Current treatment strategies focusing on pain management or surgical intervention have limited efficacy and can lead to numerous complications. Cell-based therapies involving the delivery of pro-regenerative stem cells into the intervertebral disc (IVD) hold promise as a new approach to modify the progression of the disease and promote constructive tissue repair, with the goal of improving function and reducing pain. This research project brings together a multidisciplinary research team to develop novel cell-instructive biomaterials for applications in cell delivery to promote IVD regeneration, as well as in advanced models of IVD disease. These biomaterials platforms will be designed to mimic the composition of the human IVD within a naturally-derived polymer that forms a gel at body temperature. Through detailed cell culture studies, the team will explore the proliferation and differentiation of human nucleus pulposus cells and notochord-like stem cells following encapsulation, and will tune the platform to enhance cell viability and lineage-specific differentiation to augment the regenerative potential of the cells. Future studies will extend the approach to generate tissue-specific disease models to develop a better understanding of the cellular changes that occur in IVD degeneration and the impact of the diseased microenvironment on IVD cells. Our platforms will provide much-needed insight into the effects of the local microenvironment on IVD cell function and phenotype and may be applied in future stem cell-based therapies for the treatment of IVD degeneration.
Chronic back pain associated with intervertebral disc degeneration is a debilitating condition that negatively impacts the quality of life of many Canadians. Current treatment strategies focusing on pain management or surgical intervention have limited efficacy and can lead to numerous complications. Cell-based therapies involving the delivery of pro-regenerative stem cells into the intervertebral disc (IVD) hold promise as a new approach to modify the progression of the disease and promote constructive tissue repair, with the goal of improving function and reducing pain. This research project brings together a multidisciplinary research team to develop novel cell-instructive biomaterials for applications in cell delivery to promote IVD regeneration, as well as in advanced models of IVD disease. These biomaterials platforms will be designed to mimic the composition of the human IVD within a naturally-derived polymer that forms a gel at body temperature. Through detailed cell culture studies, the team will explore the proliferation and differentiation of human nucleus pulposus cells and notochord-like stem cells following encapsulation, and will tune the platform to enhance cell viability and lineage-specific differentiation to augment the regenerative potential of the cells. Future studies will extend the approach to generate tissue-specific disease models to develop a better understanding of the cellular changes that occur in IVD degeneration and the impact of the diseased microenvironment on IVD cells. Our platforms will provide much-needed insight into the effects of the local microenvironment on IVD cell function and phenotype and may be applied in future stem cell-based therapies for the treatment of IVD degeneration.
How to make off-the-shelf cellular treatment of diabetic ulcers more effective
Investigator: Sowmya Viswanathan
Institute: University Health Network
Co-investigators: Ian Rogers, Lunenfeld-Tanenbaum Research Institute
Award Value: $75,000
 One of the major complications of diabetes is the alteration of wound healing, which can lead to chronic wounds such as diabetic foot ulcers. Diabetes prolongs the duration of normal wound healing by shifting neutrophil infiltration and monocyte/macrophages polarization towards pro-inflammatory subtypes. Mesenchymal stromal cells (MSCs) are a potential therapeutic option for diabetic wound complications. Recently, MSCs have been utilized for their anti-inflammatory functionality in Crohn’s disease, graft vs. host disease and critical limb ischemia. The Viswanathan lab has developed a novel protocol to enhance MSCs by growing them in scalable 3D suspensions in serum-free medium and proprietary cytokines or small molecules. The 3D configuration alters cell adhesion markers and results in more potent anti-inflammatory and immune modulatory MSCs without licensing or genetic modification. Importantly, the enhanced anti-inflammatory and immunomodulatory properties are maintained through cryopreservation, making 3D enhanced MSCs attractive as an off-the-shelf, ready-to use therapy for acute applications, such as diabetic ulcers. In this project, we will focus on optimizing cryopreservation parameters for 3D configurations of MSCs. Using design-of-experiment statistical approaches, we will systematically optimize combinations of parameters to cryopreserve 3D configurations of MSCs for immediate post-thaw use in a validated diabetic ulcer model in collaboration with the Rogers lab. These studies will provide proof-of-concept data for clinical trial applications using potent, ready-to use MSC products for diabetic wounds.
One of the major complications of diabetes is the alteration of wound healing, which can lead to chronic wounds such as diabetic foot ulcers. Diabetes prolongs the duration of normal wound healing by shifting neutrophil infiltration and monocyte/macrophages polarization towards pro-inflammatory subtypes. Mesenchymal stromal cells (MSCs) are a potential therapeutic option for diabetic wound complications. Recently, MSCs have been utilized for their anti-inflammatory functionality in Crohn’s disease, graft vs. host disease and critical limb ischemia. The Viswanathan lab has developed a novel protocol to enhance MSCs by growing them in scalable 3D suspensions in serum-free medium and proprietary cytokines or small molecules. The 3D configuration alters cell adhesion markers and results in more potent anti-inflammatory and immune modulatory MSCs without licensing or genetic modification. Importantly, the enhanced anti-inflammatory and immunomodulatory properties are maintained through cryopreservation, making 3D enhanced MSCs attractive as an off-the-shelf, ready-to use therapy for acute applications, such as diabetic ulcers. In this project, we will focus on optimizing cryopreservation parameters for 3D configurations of MSCs. Using design-of-experiment statistical approaches, we will systematically optimize combinations of parameters to cryopreserve 3D configurations of MSCs for immediate post-thaw use in a validated diabetic ulcer model in collaboration with the Rogers lab. These studies will provide proof-of-concept data for clinical trial applications using potent, ready-to use MSC products for diabetic wounds.
Collagen-based Capsules for Progenitor-cell Therapies
Investigator: Emilio Alarcon
Institute: University of Ottawa
Co-investigators:
Erik Suuronen, University of Ottawa Heart Institute
Darryl Davis, University of Ottawa Heart Institute
Award Value: $74,814
 Micro-encapsulation of stem cells has proven to be a practical methodology for improving cell engraftment and repair of injured heart muscle. However, to date, most microcapsules have been prepared using inert polymeric matrices and/or synthetic polymeric blends that limit its biodegradability and makes clinical translation difficult. In this project, the team will develop and assess the in vivo performance of a next generation of collagen-based microcapsules for progenitor cell therapies specifically designed to improve capsule retention and biodegradability within injured cardiac (heart) tissue. We hypothesize that micro-engineering the compositional structure of capsules, using medical grade collagen will produce materials with enhanced the length of time the cells are retained at the site of injection and biological activity within infarcted myocardium (tissues damaged by a heart attack). Since the new capsules are cleavable ; this provides a means to deliver stem cells selectively in regions where cardiac functional remodelling is taking place. Thus, our novel materials will open new avenues towards the development of bio-responsive materials for boosting functional engraftment of stem-cells within injured cardiac muscle.
Micro-encapsulation of stem cells has proven to be a practical methodology for improving cell engraftment and repair of injured heart muscle. However, to date, most microcapsules have been prepared using inert polymeric matrices and/or synthetic polymeric blends that limit its biodegradability and makes clinical translation difficult. In this project, the team will develop and assess the in vivo performance of a next generation of collagen-based microcapsules for progenitor cell therapies specifically designed to improve capsule retention and biodegradability within injured cardiac (heart) tissue. We hypothesize that micro-engineering the compositional structure of capsules, using medical grade collagen will produce materials with enhanced the length of time the cells are retained at the site of injection and biological activity within infarcted myocardium (tissues damaged by a heart attack). Since the new capsules are cleavable ; this provides a means to deliver stem cells selectively in regions where cardiac functional remodelling is taking place. Thus, our novel materials will open new avenues towards the development of bio-responsive materials for boosting functional engraftment of stem-cells within injured cardiac muscle.
Characterization of immune cells during human pancreatic development
Investigator: Maria Cristina Nostro
Institute: University Health Network
Co-investigators: Gordon Keller, University Health Network
Award Value: $75,000
 There are two forms of diabetes mellitus: type 1 diabetes (T1D), characterized by the destruction of insulin-producing cells (β cells) by the immune system and type 2 diabetes (T2D), a metabolic disorder that is linked to insulin resistance and β cell failure. Currently, T1D is treated by insulin administration using needle injections or pumps. Patients with T2D are treated with medications that increase insulin secretion from the β cells, reduce sugar production by the liver or enhance insulin sensitivity. Despite these treatments, poorly controlled sugar levels in the blood leads to severe complications, including blindness, limb amputations, kidney failure and cardiovascular diseases. According to Statistics Canada, diabetes mellitus is the 6th leading cause of death in Canada. It is clear that in order to improve these figures, new generations of therapies will be required. The recent progress in stem cell research is bringing us closer to developing β cell therapies for T1D. This proposal leverages on Drs. Nostro and Keller’s expertise on the differentiation of insulin-producing pancreatic β cells and hematopoietic cells. Together, we propose to profile various cell types in the developing human pancreas to identify novel regulatory mechanisms that control β cell maturation and use this information to optimize the production of β cells from human pluripotent stem cells (hPSCs), generating a cell type that has potential for transplantation into patients with T1D.
There are two forms of diabetes mellitus: type 1 diabetes (T1D), characterized by the destruction of insulin-producing cells (β cells) by the immune system and type 2 diabetes (T2D), a metabolic disorder that is linked to insulin resistance and β cell failure. Currently, T1D is treated by insulin administration using needle injections or pumps. Patients with T2D are treated with medications that increase insulin secretion from the β cells, reduce sugar production by the liver or enhance insulin sensitivity. Despite these treatments, poorly controlled sugar levels in the blood leads to severe complications, including blindness, limb amputations, kidney failure and cardiovascular diseases. According to Statistics Canada, diabetes mellitus is the 6th leading cause of death in Canada. It is clear that in order to improve these figures, new generations of therapies will be required. The recent progress in stem cell research is bringing us closer to developing β cell therapies for T1D. This proposal leverages on Drs. Nostro and Keller’s expertise on the differentiation of insulin-producing pancreatic β cells and hematopoietic cells. Together, we propose to profile various cell types in the developing human pancreas to identify novel regulatory mechanisms that control β cell maturation and use this information to optimize the production of β cells from human pluripotent stem cells (hPSCs), generating a cell type that has potential for transplantation into patients with T1D.
2018-2019 Awardees:
- Elucidation of splicing-based mechanisms underlying cell fate control that represent targets in regenerative medicine applications ($75,000). Project Leader: Benjamin Blencowe (University of Toronto)
- A matter of timing: how the circadian clock regulates intestinal regeneration ($75,000). Project Leader: Phillip Karpowicz (University of Windsor)
- Developing new reprogramming strategies for cell replacement therapy of glaucoma ($75,000) Project Leader: Pierre Mattar (Ottawa Hospital Research Institute)
- Making new neurons from resident brain cells: a new therapy for stroke repair? ($74,946) Project Leader: Maryam Faiz (University of Toronto)
- Collaborative laboratory-based study of stem cell therapies: Preclinical Multicenter Acute lung injury Trials In Canada (PreMATIC) ($74,997) Project Leader: Manoj Lalu (Ottawa Hospital Research Institute)
- Boosting cellular energy signals in muscle stem cells as a therapy for muscular dystrophy ($75,000) Project Leader: Keir Menzies (University of Ottawa)
2017-2018 Awardees (*co-funded by Medicine by Design; **Funded by Medicine by Design):
- The impact of environmental pollutants on pancreas development ($75,000) Project Leader: Jennifer Bruin (Carleton University)
- A 3D model of muscle to study potential therapies for Duchenne muscular dystrophy* ($75,000) Project Leader: Penney M. Gilbert (University of Toronto)
- Tailoring donor lungs to control immune response before transplant** ($75,000) Project Leader: Stephen C. Juvet (University Hospital Network)
- Improving the creation of bile duct cells to model liver disease** ($75,000) Project Leader: Binita M. Kamath (SickKids Research Institute)
- A computational modelling platform to support imaging and tissue design** ($75,000) Project Leader: John Parkinson (SickKids Research Institute)
- Understanding radial glial cell response to neural injury at a single cell level** ($75,000) Project Leader: Bret Pearson (SickKids Research Institute)
- A therapeutic strategy for treating Duchenne Muscular Dystrophy ($75,000) Project Leader: Michael A. Rudnicki (Ottawa Hospital Research Institute)
- Magnetic resonance imaging to assess stem cell treatments for lung diseasel** ($75,000) Project Leader: Giles Santyr (SickKids Research Institute)
- Autism spectrum disorder drug testing using human neurons ($75,000) Project Leader: Karun Singh (McMaster University)
- Clinical investigation of cell therapy to treat age-related osteoporosis ($75,000) Project Leader: William L Stanford (Ottawa Hospital Research Institute)
- Making new blood vessels for life-threatening lung diseases in newborns ($75,000) Project Leader: Bernard Thébaud (Ottawa Hospital Research Institute)
- New ways to stimulate the production of insulin-producing cells to treat diabetes* ($75,000) Project Leader: Michael B. Wheeler (University of Toronto)
Past Awardees (*co-funded by Medicine by Design):
- Control of immune tolerance toward allograft cell transplants ($50,000) Project Leader: Andras Nagy (The Lunenfeld-Tanenbaum Research Institute) 2015-16
- Dissolving scar tissue in spinal cord injuries improve candidacy and effects of stem cell transplants* ($49,988) Project Leader: Charles Tator (University Health Network) 2015-16
- Sole fuel source to enhance pluripotency ($50,000) Project Leader: Dean Betts (Western University) 2015-16
- An injectable biomaterial system for the delivery of stem cells in the treatment of retinal disease ($50,000) Project Leader: Heather Sheardown (McMaster University) 2015-16
- Activating enhancers to improve reprogramming efficiency* ($50,000) Project Leader: Jennifer Mitchell (University of Toronto) 2015-16
- Better maturation of iPS-derived heart cells ($49,200) Project Leader: John Coles (SickKids Research Institute) 2015-16
- Protein inactivation by agrochemicals as a mechanism underlying development of Autism Spectrum Disorder ($49,994) Project Leader: John Vessey (University of Guelph) 2015-16
- Injectable, tissue engineered scaffold for delivery of cardiac patches* ($50,000) Project Leader: Milica Radisic (University of Toronto) 2015-16
- Heart tissue repair via immune cell growth factors* ($50,000) Project Leader: Slava Epelman (University Health Network) 2015-16
- Intestinal stem cells and gut microbiota in early postnatal development and necrotizing enterocolitis ($49,980) Project Leader: Tae-Hee Kim* (SickKids Research Institute) 2015-16
- Biomimetic surfaces for directed differentiation of lung stem cells * ($49,983) Project Leader: Tom Waddell (Univerisity Health Network) 2015-16
- Mechanisms regulating differentiation in hemangioma stem cells ($49,500) Project Leader: Zia Khan (Western University) 2015-16
- TIMP-engineered niches for liver progenitor cell expansion ($75,000) Project Leader: Rama Khokha (University Health Network) 2014-15
- Control of RNA translation into proteins in human stem cells, neurons and disease ($75,000) Project Leader: James Ellis (SickKids Research Institute) 2014-15
- Engineering a functional human thymus from pluripotent stem cells ($75,000) Project Leader: Peter Zandstra (University of Toronto) 2014-15
- Role of short RNA fragments in mediating the anti-inflammatory effects of bone marrow stem cells in sepsis ($75,000) Project Leader: Duncan Stewart (Ottawa Hospital Research Institute) 2014-15
- Genomic correction of cardiac sarcomeric protein mutations in iPSC-derived cardiomyocytes ($75,000)Project Leader: John Coles (SickKids Research Institute) 2014-15
Other Current Funding:
Disease Teams
Fellowships
Since 2014, OIRM has invested more than $14 million in promising research aimed at answering key questions to advance stem cells towards new treatments and products for the health and economic benefit of Ontarians.
With our help, Ontario’s researchers are delivering results, including:
- OIRM researcher Lauralyn McIntyre at The Ottawa Hospital published the results of the world’s first clinical trial using mesenchymal stem/stromal cells to treat septic shock, a life-threatening condition that can occur when an infection spreads throughout the body. OIRM is supporting a randomized, multi-site trial to begin in 2018 across Canada. Read about one of the patients in the phase 1 trial. (2017)
- The Disease Team research of OIRM scientists Michael Laflamme and Gordon Keller, University Health Network, became a foundational part of BlueRock Therapuetics’ initial development. His research, with co-investigator Gordon Keller, is an innovative method to regenerate heart cells after a heart attack, which could substantially increase quality of life and reduce long-term health care costs for patients. OIRM invested more than $2 million in direct research costs. (2016) Read more about this research.
- OIRM researchers at the Ottawa Hospital, led by Mark Freedman and Harry Atkins, published results of a long-term study showing that aggressive multiple sclerosis can be completely suppressed, and possibly even reversed, with a blood stem cell transplant. Patients who received this treatment experienced a halt to disease progression for a prolonged period of time, without the use of disease-modifying drugs. Read the scientific paper published in The Lancet. Read more about this clinical trial. (2016)
- An early phase study led by OIRM researcher Harry Atkins at the Ottawa Hospital demonstrated that blood stem cell transplantation led to remission in patients with severe myasthenia gravis, a potentially life-threatening autoimmune disease that affects the muscles. Patients experienced consistent, durable, symptom- and treatment-free remission. This study was published in JAMA Neurology. (2016)
- OIRM investigator Dr. Terry Yau at the University Health Network (UHN) in Toronto showed that stem cell therapy during coronary artery bypass grafting for heart disease is safe and feasible for improving heart function in patients. This promising advance will now move to larger clinical studies. Published in the Journal of Thoracic and Cardiovascular Surgery. (2016)
- Results of a phase one study led by Dr. Duncan Stewart at the Ottawa Hospital showed safety of genetically-enhanced stem cells as a potential therapy for pulmonary arterial hypertension, a severe lung disease. Published in Circulation Rsearch (2015), this study will begin phase two in several centres across Canada in 2018.
OIRM has over 250 member scientists in nine centres across the province, including stem cell biologists, biomaterial engineers and translational clinicians.
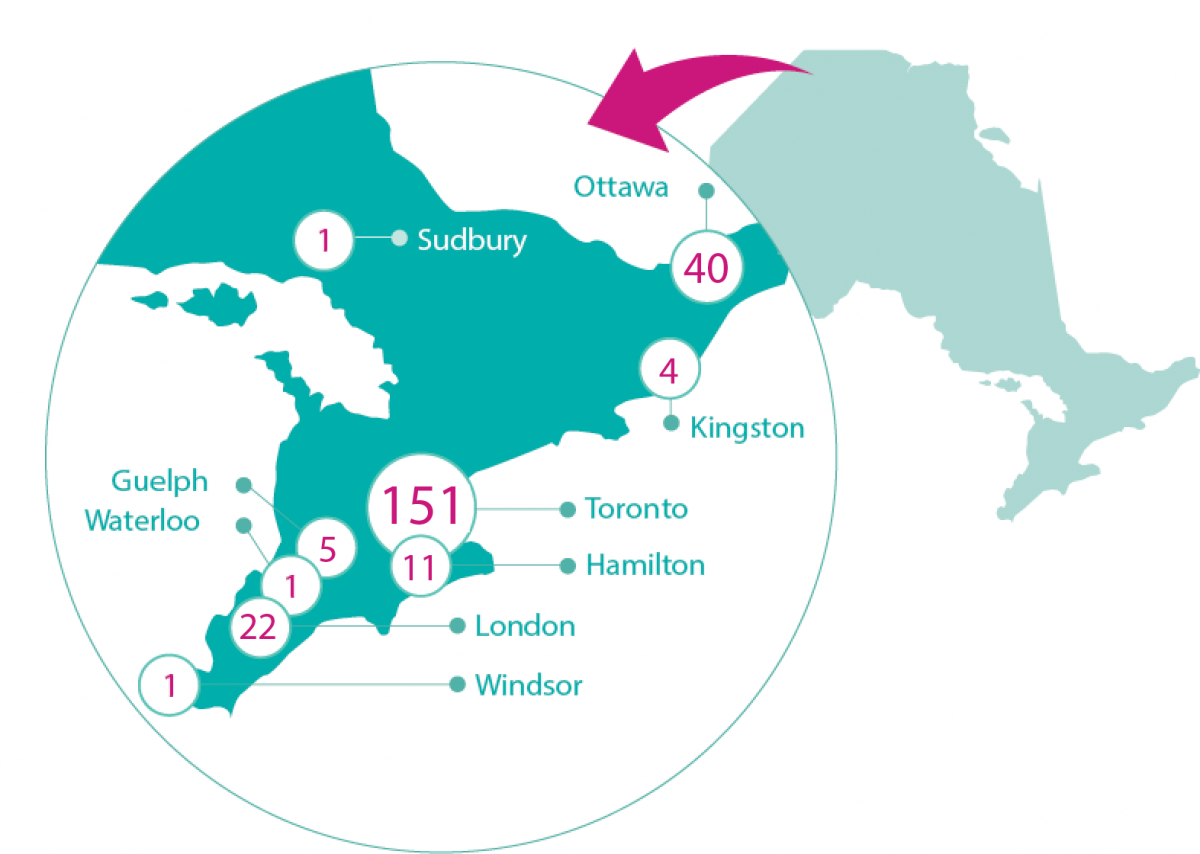
We support our researchers and their trainees in their quest to answer key questions that advance stem cell research towards improved health for patients around the world. OIRM embraces collaboration, and its research and training portfolio is centred around building effective partnerships to best leverage our funding. This approach allows us to support a larger number of research projects and clinical trials. View our current list of funded research projects.
Opportunities within OIRM’s research and training program include the following:
Research & Training Funding
Partnerships & Industry Offers
Clinical Program & Manufacturing Resources
Workshops, Courses & Events
Career Opportunities
OIRM Membership
OIRM’s research program includes large, disease-based team grants, smaller New Ideas Grants to answer basic research questions and Post-doctoral Fellowships.
2019-2020 Disease Team Awardees:
A trial to improve wound healing using stem cells from patient’s own discarded burned tissue.
Investigator: Marc Jeschke
Institute: Sunnybrook Research Institute
Award Value: $450,000
 The single most important factor that determines survival of a burn patient is wound healing. This team recently isolated cells identified as burn-derived mesenchymal stem cells (BD-MSCs) from discarded burned skin. A skin substitute, Integra®-SC, was engineered by incorporating BD-MSCs into Integra®, a dermal matrix, and found beneficial results in both small and large animal models. In this study the team will conduct a first-in-humans clinical trial using Integra®-SC, developed from a patient’s own surgically removed burned tissue. It’s believed that Integra®-SC will facilitate and improve wound healing, heal faster, and, in the long-term, result in less scar formation. This approach also reduces the burden of surgeons needing to surgically remove a patient’s own good uninjured skin to use as a donor. This study will be conducted in stages followed by interim safety analyses. After safety analyses, the team will evaluate any needs or gaps in order to include recruitment of patients with a larger percent total body surface area burn. Integra®-SC has a broad clinical application and will impacts the larger burn community, patients with traumatic and complex wounds, and the stem cell research community. The clinical applicability of these results is promising and could create a new standard for the way we care for patients in the province of Ontario and world-wide.
The single most important factor that determines survival of a burn patient is wound healing. This team recently isolated cells identified as burn-derived mesenchymal stem cells (BD-MSCs) from discarded burned skin. A skin substitute, Integra®-SC, was engineered by incorporating BD-MSCs into Integra®, a dermal matrix, and found beneficial results in both small and large animal models. In this study the team will conduct a first-in-humans clinical trial using Integra®-SC, developed from a patient’s own surgically removed burned tissue. It’s believed that Integra®-SC will facilitate and improve wound healing, heal faster, and, in the long-term, result in less scar formation. This approach also reduces the burden of surgeons needing to surgically remove a patient’s own good uninjured skin to use as a donor. This study will be conducted in stages followed by interim safety analyses. After safety analyses, the team will evaluate any needs or gaps in order to include recruitment of patients with a larger percent total body surface area burn. Integra®-SC has a broad clinical application and will impacts the larger burn community, patients with traumatic and complex wounds, and the stem cell research community. The clinical applicability of these results is promising and could create a new standard for the way we care for patients in the province of Ontario and world-wide.
Towards a cell therapy application using pluripotent stem cell-derived heart muscle cells to regenerate injured hearts.
Investigator: Michael Laflamme
Institute: University Health Network
Co-investigators:
Graham Wright, Sunnybrook Research Institute
Gordon Keller, University Health Network
Ren-Ke Li, University Health Network
Terrence Yau, University Health Network
Award Value: $450,000

After myocardial infarction (heart attack), the heart muscle lost is replaced by non-contractile scar tissue, often initiating progressive heart failure. The current options for treating post-infarct heart failure are limited, and this remains a disease with high morbidity, mortality and societal costs. The ability to “remuscularize” the infarct zone by transplanting cardiomyocytes (heart muscle cells) derived from human pluripotent stem cells (hPSCs) represents a potentially revolutionary new therapy for patients suffering from this disease. The team has successfully translated protocols for the cardiogenic differentiation of hPSCs from the lab bench to large-scale production, and are now able to routinely generate hPSC-derived cardiomyocytes in batches of >1×109 cells. In addition, the team has improved protocols that guide the differentiation of hPSCs into specialized cardiac subtypes, including the ventricular cardiomyocytes that are needed for infarct repair, and have recently partnered with a new Toronto-based biotechnology company, BlueRock Therapeutics, that will further upscale and help commercialize these technologies. This project builds on these advances and tackles critical next steps including proof-of-concept work to determine if the transplantation of hPSC-derived ventricular myocytes can remuscularize the infarct scar and improve contractile function in well-validated models of post-infarct heart failure. The latter study will thoroughly test the safety and efficacy of this novel cell therapy using endpoints including histology (host and graft tissue structure), MRI (contractile function), as well as ECG recording and mapping studies (electrical function). With the successful completion of this preclinical work, the team hopes to launch an Ontario-based, first-in-humans trial of hPSC-derived ventricular myocytes as a cell-based therapy for heart failure.
Stimulating muscle repair for muscular dystrophy
Investigator: Michael A. Rudnicki
Institute: Ottawa Hospital Research Institute
Co-investigators:
Penney Gilbert, University of Toronto
Patrick Gunning, University of Toronto
William Stanford, Ottawa Hospital Research Institute
Jodi Chardon, The Ottawa Hospital
Hugh McMillian, Children’s Hospital of Eastern Ontario
Award Value: $300,000

Duchenne muscular dystrophy (DMD) is a devastating genetic disease that effects 1 in 3,500 boys that is characterized by progressive muscle wasting and premature death in the second or third decade of life. No effective cure-based therapy exists for DMD. This multidisciplinary team is implementing innovative therapeutic approaches for treating DMD patients to extend physical mobility and enhance quality of life by stimulating skeletal muscle regeneration using small molecule drugs. Previous studies have identified a panel of small molecule drugs that that have identified target pathways that can be modulated to ameliorate the regenerative defect in DMD muscle stem cells. Candidate drugs will be tested on dystrophic models and pathways validated in human muscle stem cells to identify the best candidates to advance towards the clinic. The team expects that these drugs could be combined with other therapies, such as exon skipping or gene therapy, to provide a synergistic approach to treatment. Utilizing rigorous preclinical and clinical trial methodology design will support a pathway to first-in-human clinical studies. The project is partnered with Satellos Bioscience Inc.
INCuBATOR: New Cell Treatments for Lung Injury in Babies – Getting research faster and safer into patients
Investigator: Bernard Thébaud
Institute: Ottawa Hospital Research Institute
Co-investigators:
Dean Fergusson, Ottawa Hospital Research Institute
Justin Presseau, Ottawa Hospital Research Institute
Kednapa Thavorn, Ottawa Hospital Research Institute
Award Value: $300,000

Extreme prematurity is the main cause of mortality and morbidity in children before five years of age. The most severe complication is bronchopulmonary dysplasia (BPD), a chronic lung disease that follows ventilator and oxygen treatment for acute failure to breathe. BPD also adversely affects the brain and leads to blindness. Currently, there is no treatment for BPD. Because these injuries occur in developing organs, consequences are life-long and carry a high economic burden. Thus, effective interventions at this stage of life provide exceptional value. This team has shown that umbilical cord-derived mesenchymal stromal cells (UC-MSC) repair lung injury in neonatal rodents modelling BPD. UC has numerous advantages over bone marrow as a source of MSCs, especially for newborn disease. As it is easy to obtain after birth without harming mother or baby; cells have higher MSC yield and expansion potential as well as higher repair capabilities. The team has developed a highly efficient, closed and fully-automated system that yields high numbers of low passage, GMP manufactured clinical grade UC-MSCs. The INCuBATOR approach will expedite the process of bringing this promising cell therapy into patients through a multi-disciplinary, rigorous and efficient process that will mitigate the potential high-risk nature of cell therapy in newborns. This is supported through unprecedented access to a large animal model, that mimics the human disease to test the safety, efficacy and immune response of UC-MSC. Rigorous analyses (systematic reviews) of all studies testing MSCs in animal models of BPD and of clinical studies testing MSCs in preterm infants will help to inform preclinical and clinical design, leading to the delivery of a Phase I study protocol to test the feasibility and safety of UC-MSCs in preterm infants at risk of developing BPD .
Cell transplantation to preserve central vision
Investigator: Valerie Wallace
Institute: University Health Network
Co-investigators:
Andras Nagy, Lunenfeld-Tanenbaum Research Institute
Carol Schuurmans, SunnyBrook Research Institute
Molly Shoichet, University of Toronto
Derek van der Kooy, University of Toronto
Award Value: $300,000

Late stage retinitis pigmentosa (RP) and age‐related macular degeneration (AMD) are degenerative diseases that rob patients of central high acuity vision. The multiple underlying etiologies of these diseases ultimately converge on the loss of light sensitive cone photoreceptors in the central macular region of the retina, resulting in central vision loss. To date, there are few experimental treatments that preserve cone photoreceptor function and survival. Based on clinical trial results, neurotrophic factor delivery is not therapeutically beneficial, prompting the need to develop alternate neuroprotective approaches. Rod photoreceptors and RPE are essential for the survival and function of cone photoreceptors and, therefore, this study will exploit the paracrine effects of these cells to promote cone survival. To maximize the paracrine effects of rods and RPE this study will use combinatorial cell therapy to preserve cones in animal models of secondary cone degeneration, an approach that will be generally applicable to the maintenance of central vision in late stage RP and AMD. The team will develop an optimized procedure for the efficient induction and enrichment of human rod photoreceptors and RPE from stem cells and fibroblasts using small molecules and transcription factor reprogramming strategies, develop and test novel biomaterials enhanced with photoreceptor survival factors, compare the survival and functional engraftment potential of single and combinatorial rod/RPE delivery on cone survival and function in animal models of late stage RP, and provide solutions for cell therapy safety and allograft immune tolerance.
Cellular immunotherapy for septic shock (CISS): Research to move stem cells through the clinical pipeline
Investigator: Lauralyn McIntyre
Institute: Ottawa Hospital Research Institute
Co-investigators:
Shirley Mei, Ottawa Hospital Research Institute
Kednapa Thavorn, Ottawa Hospital Research Institute
Claudia dos Santos, St. Michael’s Hospital
Jason Acker, Canadian Blood Services
Dean Fergusson, Ottawa Hospital Research Institute
Award Value: $200,000

Septic shock is a devastating illness and the most severe form of infection seen in the intensive care unit (ICU). Approximately 20 – 40% of those with septic shock will die and survivors suffer significant long-term impairment in function and reduced quality of life. Despite decades of research examining different therapies, none has proven successful and supportive care remains the mainstay of therapy, at a cost of approximately $4B in Canada annually. Mesenchymal stem cells (MSCs) represent a novel treatment, and have been shown to modulate the immune system, increase clearance of bacterial pathogens, restore organ function, and reduce death in preclinical models. Over the last seven years, this multi-disciplinary team has taken the lead to develop a research program studying MSCs in septic shock, and was the first in the world to have conducted and completed a Phase I clinical trial that evaluated MSCs in patients with septic shock. This trial established that MSCs appear safe and that a randomized controlled trial is feasible. Based on these results, we are now moving to a larger clinical trial at several academic hospitals across Ontario and Canada. This Phase II RCT (CISS2) will continue to evaluate safety, assess if there are strong signals for clinical benefit, and examine mechanisms and biological effects of MSCs. An economic analysis will also determine if the treatment is co
st effective. This team continues to optimize protocols for cryopreservation of MSC products, identify bio-signatures tailored to septic shock to further identify potent cells, and to develop MSC modifications to further improve their performance characteristics for evaluation in future septic shock clinical trials.
Stem cell approaches to repairing damaged white matter in the brain of children and teenagers
Investigator: E. Ann Yeh
Institute: The Hospital for Sick Children
Co-investigators:
Freda Miller, The Hospital for Sick Children
Donald Mabbott, The Hospital for Sick Children
Paul Frankland, The Hospital for Sick Children
David Kaplan, The Hospital for Sick Children
Cindi Morshead, University of Toronto
Douglas Munoz, Queen’s University
Jing Wang, Ottawa Hospital Research Institute
Award Value: $200,000
 Damage to the white matter of the brain occurs following injury and in disorders like multiple sclerosis. This results in sensory, motor, and cognitive problems. Currently, there are no effective medical therapies to promote brain repair and reduce disability following white matter damage. This project looks at enhancing the genesis of new oligodendrocytes (the cells that make myelin) and in doing so, promote white matter repair. Taking advantage of the fact that our brains contain resident precursor cells that normally make oligodendrocytes throughout life, the team will ask whether endogenous precursors can be pharmacologically activated to promote repair. Excitingly, this team has discovered that a widely-used and safe drug, metformin, will enhance the genesis of oligodendrocytes from neural precursors and that, following pediatric neural injury, this promotes neuroanatomical and functional recovery. This work will now be translated to the clinic. The team is pursuing a pilot clinical trial of metformin for pediatric demyelinating disease using outcome measures recently developed. Positive results from this clinical trial will lead to a dramatic shift in how we treat children/teenagers with white matter injury, and will pave the way for future additional clinical trials in children and adults with white matter damage.
Damage to the white matter of the brain occurs following injury and in disorders like multiple sclerosis. This results in sensory, motor, and cognitive problems. Currently, there are no effective medical therapies to promote brain repair and reduce disability following white matter damage. This project looks at enhancing the genesis of new oligodendrocytes (the cells that make myelin) and in doing so, promote white matter repair. Taking advantage of the fact that our brains contain resident precursor cells that normally make oligodendrocytes throughout life, the team will ask whether endogenous precursors can be pharmacologically activated to promote repair. Excitingly, this team has discovered that a widely-used and safe drug, metformin, will enhance the genesis of oligodendrocytes from neural precursors and that, following pediatric neural injury, this promotes neuroanatomical and functional recovery. This work will now be translated to the clinic. The team is pursuing a pilot clinical trial of metformin for pediatric demyelinating disease using outcome measures recently developed. Positive results from this clinical trial will lead to a dramatic shift in how we treat children/teenagers with white matter injury, and will pave the way for future additional clinical trials in children and adults with white matter damage.
Past Awardees:
- A stem cell approach to regenerate the injured spinal cord ($400,000) Project Leader: Michael Fehlings (University Health Network)
- Cellular Immunotherapy for Septic Shock ($400,000) Project Leader: Duncan Stewart (Ottawa Hospital Research Institute)
- Towards a cell therapy application for using pluripotent stem cell-derived cardiomyocytes to treat cardiovascular disease ($250,000) Project Leader: Gordon Keller (University Health Network) –
- Cone photoreceptor derivation and transplantation – an innovative approach for the treatment of age-related macular regeneration ($250,000) Project Leader: Valerie Wallace (University Health Network) –
- Application of novel stem cell derived human non-monocytic dendritic cell precursors (hNM-DCPs) for immunotherapies ($250,000) Project Leader: Mick Bhatia (McMaster University)
- Preclinical evaluation of a bioengineered human islet ($100,000) Project Leader: Cristina Nostro (University Health Network)
- Overcoming central vision loss with stem cell therapy and rehabilitation ($100,000) Project Leader: Valerie Wallace (University Health Network
- Pluripotent and cord blood derived progenitor t-cells for immune reconstitution therapy ($100,000) Project Leader: JC Zuniga-Pflucker (Sunnybrook Research Institute)
Other Current Funding:
New Ideas Grants
Fellowships
From 2014 – 2020 OIRM supported four research-based programs for Ontario-based researchers and/or trainees.
As of 2020, OIRM is no longer able to provide research funding.
OIRM Disease Team projects encompass large, collaborative research teams at multiple institutions across Ontario with potential partnerships outside the province. Disease Team grants support projects that are developing novel cellular or stem cell-related therapeutic approaches to tissue repair and regeneration for specific diseases. All OIRM Disease Teams are aimed at developing therapies with a clear path to the clinic. Grants were issued on an annual basis following a competitive, externally-reviewed process and are valued up to $750,000.
OIRM’s New Ideas program supports projects at the discovery level in the area of stem cell and regenerative medicine research. New Ideas projects are typically based at one institution with one or two investigators. The projects have clear and achievable short-term goals leading to clinical or commercial activities. Grants were issued on an annual basis following a competitive, externally-reviewed process and are valued up to $75,000.
The OIRM Fellowship Award supported outstanding trainees in basic research or health professions for up to three years to pursue a specific line of research in bioengineering, stem cells and/or regenerative medicine.
Trainee travel awards are available for qualifying applicants for OIRM supported workshops, events and conferences.
Non-OIRM funding opportunities are also posted to help support the research community.
Funding Opportunities
New Idea Grants
Currently, there are no plans for a New Idea Grants competition.
Disease Team Grants
Currently, there are no plans for a Disease Team Grant competition.
OIRM Fellowship Award
Currently closed.
Trainee Travel Awards
Non-OIRM Funding Opportunities
OIRM is a network of 1700+ stem cell biologists, bioengineers, clinicians, industry and business leaders and their trainees working within stem cell and regenerative medicine in Ontario. With members located in nine cities across the province, we are a community that supports collaboration between labs and across disciplines to advance both basic research and clinical translation.
Membership Criteria
- Members must hold a primary, full-time faculty appointment at an Ontario University or affiliated Research Institute
- Members require a PhD and/or MD in a scientific field appropriate to stem cell and regenerative medicine research
- Members require at least two published academic papers in the field of stem cell and regenerative medicine; for clinicians, a strong interest in regenerative medicine approaches in their area of clinical expertise can act as a substitute
Membership Benefits
As a member of our community, you and your trainees have access to many resources and programs to help support your research. OIRM member benefits include:


You are an important member of this community and as a member of OIRM, we encourage you be active members of the community, through your own participation in OIRM activities and by encouraging your trainees to attend our education talks and workshops. Because the strength of the community is built upon its scientists, OIRM members may be called upon to provide talks, engage with the public and become advocates for the OIRM’s programs and opportunities. Download our membership benefits document.
Membership Application
If you meet the above criteria and are willing to be an active part of our community, please provide the following documentation to jodi.garner@oirm.ca
- A one-paragraph statement on official letterhead explaining why you would like to be a member of OIRM and how our network can benefit your research
- Updated CV, highlighting publications in the field of stem cell and regenerative medicine
After an internal review, those approved, will be added to our “OIRM Scientists” webpage. To complete your profile you will be asked to provide the following information;
- link to your university profile
- link to your hospital/research institute profile
- one paragraph on your research interests
- link to your PubMed listing
- five key words to describe your research
- a high-resolution photo (head shot)
- admin contact info (if applicable)
- a list of trainees and email addresses
For more information about OIRM membership, our membership benefits or to apply please contact Jodi.garner@oirm.ca.
 Adult skeletal muscle has the remarkable capacity to regenerate in response to injury, which is imparted by muscle stem cells (MuSCs). Even though adult skeletal muscle is a diverse tissue comprised of different muscle groups with various fiber type compositions, our understanding of how a MuSC makes a myofiber is a linear process with sequential activation of transcription factors. This prevailing dogma of linear activation during MuSC commitment cannot account for the functional diversity observed at the tissue level or the bipotential activity of MuSCs, such as making brown fat. We hypothesize that MuSCs are primed and exist in discrete subpopulations that differentiate down multiple lineages. The existence of subpopulations suggests population heterogeneity of the MuSCs and a hierarchical organization with distinct functional properties. To address this our work will combine classical lineage tracing experiments with molecular profiling and characterization. Use of single cell RNA sequencing will help to unravelling the complexities by profiling single cells instead of heterogeneous bulk populations. We hypothesize that using scRNAseq of MuSCs in combination with lineage tracing models; will uncover novel subpopulations and their regulators, and ultimately, deconstruct the myogenic lineages.
Adult skeletal muscle has the remarkable capacity to regenerate in response to injury, which is imparted by muscle stem cells (MuSCs). Even though adult skeletal muscle is a diverse tissue comprised of different muscle groups with various fiber type compositions, our understanding of how a MuSC makes a myofiber is a linear process with sequential activation of transcription factors. This prevailing dogma of linear activation during MuSC commitment cannot account for the functional diversity observed at the tissue level or the bipotential activity of MuSCs, such as making brown fat. We hypothesize that MuSCs are primed and exist in discrete subpopulations that differentiate down multiple lineages. The existence of subpopulations suggests population heterogeneity of the MuSCs and a hierarchical organization with distinct functional properties. To address this our work will combine classical lineage tracing experiments with molecular profiling and characterization. Use of single cell RNA sequencing will help to unravelling the complexities by profiling single cells instead of heterogeneous bulk populations. We hypothesize that using scRNAseq of MuSCs in combination with lineage tracing models; will uncover novel subpopulations and their regulators, and ultimately, deconstruct the myogenic lineages.
 Damaged neurons in the adult human central nervous system (CNS) do not readily regenerate. This lack of regeneration limits the functional recovery for CNS injuries such as stroke, traumatic brain and spinal cord injuries, multiple sclerosis, infection and other related conditions that involve axon pathology. The lack of ability for human neurons to regrow their axons is a major therapeutic limitation. However, recent animal studies suggest that lack of axon regeneration is the result of intrinsic inhibitory signals (blocking signals) that can be neutralized. These findings raise the exciting prospect that human CNS neurons can be coaxed into regenerating their axons if the inhibitory blockades can be identified and removed. In this project, a genome-wide search for all intrinsic inhibitory factors that prevent axon regeneration will be conducted. This will be accomplished by establishing an in vitro axon injury and regeneration assay using CNS neurons derived from human pluripotent stem cells and will be subjected to a genome-wide CRISPR knockout screen. This project seeks to identify genes which when deleted, allow the human axons to regenerate. This research will establish a genome-wide approach to study the genetic regulation of human axon regeneration, and identify targets for potential therapeutic intervention for a wide spectrum of CNS diseases that involve axon pathology.
Damaged neurons in the adult human central nervous system (CNS) do not readily regenerate. This lack of regeneration limits the functional recovery for CNS injuries such as stroke, traumatic brain and spinal cord injuries, multiple sclerosis, infection and other related conditions that involve axon pathology. The lack of ability for human neurons to regrow their axons is a major therapeutic limitation. However, recent animal studies suggest that lack of axon regeneration is the result of intrinsic inhibitory signals (blocking signals) that can be neutralized. These findings raise the exciting prospect that human CNS neurons can be coaxed into regenerating their axons if the inhibitory blockades can be identified and removed. In this project, a genome-wide search for all intrinsic inhibitory factors that prevent axon regeneration will be conducted. This will be accomplished by establishing an in vitro axon injury and regeneration assay using CNS neurons derived from human pluripotent stem cells and will be subjected to a genome-wide CRISPR knockout screen. This project seeks to identify genes which when deleted, allow the human axons to regenerate. This research will establish a genome-wide approach to study the genetic regulation of human axon regeneration, and identify targets for potential therapeutic intervention for a wide spectrum of CNS diseases that involve axon pathology. Progressive supranuclear palsy (PSP) is a neurodegenerative disease affecting mobility, behavior, speech, and thinking. The brain of a PSP patient contains many dead or dying brain cells with internal protein aggregates. The main component of these aggregates is a protein called Tau, which, in a healthy brain, supports vital cell transport systems inside the brain cells. However, Tau aggregates in patients with PSP cause brain cell dysfunction and death. Currently, there is no medication for the core symptoms of PSP.
Progressive supranuclear palsy (PSP) is a neurodegenerative disease affecting mobility, behavior, speech, and thinking. The brain of a PSP patient contains many dead or dying brain cells with internal protein aggregates. The main component of these aggregates is a protein called Tau, which, in a healthy brain, supports vital cell transport systems inside the brain cells. However, Tau aggregates in patients with PSP cause brain cell dysfunction and death. Currently, there is no medication for the core symptoms of PSP. Congenital myopathies and muscular dystrophies (CMDs) are heterogeneous disorders, many of which have neurodevelopmental co-morbidities. Defects in brain development, such as cerebral hypoplasia, are often considered secondary to the primary muscle disease phenotype. Previous research has discovered that movement is critical for promoting normal brain development (neurogenesis) and growth of the post-embryonic forebrain. Restricting movement caused premature differentiation of neural progenitors and brain dysfunction. Movement-dependent forebrain neurogenesis requires somatosensory feedback from the body. Such a relationship between movement and brain development might explain the known co-morbidity between physical and neurological deficits in congenital muscle diseases (CMDs). We propose to characterize the post-embryonic forebrain growth in established models of CMD and test the possibility of combatting aberrant neurodevelopment by providing sensorimotor input to the brain. To do so, we will characterize the changes underlying neurogenesis leading to a reduction in forebrain growth in selected CMD mutants. Second, we will test if aberrant brain development associated with CMD can be combatted via targeted activation of dorsal root ganglion (DRG) neurons and finally, we will examine whether norepinephrine is a key mediator of sensorimotor dependent forebrain neurogenesis. This research will reveal the neurodevelopmental consequences of CMD and provide novel therapeutic avenues for promoting healthy brain growth in CMD patients.
Congenital myopathies and muscular dystrophies (CMDs) are heterogeneous disorders, many of which have neurodevelopmental co-morbidities. Defects in brain development, such as cerebral hypoplasia, are often considered secondary to the primary muscle disease phenotype. Previous research has discovered that movement is critical for promoting normal brain development (neurogenesis) and growth of the post-embryonic forebrain. Restricting movement caused premature differentiation of neural progenitors and brain dysfunction. Movement-dependent forebrain neurogenesis requires somatosensory feedback from the body. Such a relationship between movement and brain development might explain the known co-morbidity between physical and neurological deficits in congenital muscle diseases (CMDs). We propose to characterize the post-embryonic forebrain growth in established models of CMD and test the possibility of combatting aberrant neurodevelopment by providing sensorimotor input to the brain. To do so, we will characterize the changes underlying neurogenesis leading to a reduction in forebrain growth in selected CMD mutants. Second, we will test if aberrant brain development associated with CMD can be combatted via targeted activation of dorsal root ganglion (DRG) neurons and finally, we will examine whether norepinephrine is a key mediator of sensorimotor dependent forebrain neurogenesis. This research will reveal the neurodevelopmental consequences of CMD and provide novel therapeutic avenues for promoting healthy brain growth in CMD patients. The human heart rate is controlled by two specialized pacemaker tissues in the heart known as the sinoatrial node (SAN) and the atrioventricular node (AVN). The SAN initiates electrical signals that drive the heartbeat while the AVN is responsible for propagating these electric signals from the upper heart chambers to the bottom heart chambers. This activates their contraction and results in the pumping of blood through the body. Failure of either pacemaker tissue leads to a life-threatening irregular or slow heartbeat and needs to be treated by implantation of an electronic pacemaker device. Approximately 21,000 Canadians receive an electronic pacemaker every year. While representing an effective treatment, electronic pacemakers have a number of disadvantages including the risk of pacemaker lead infections, the need for surgical battery replacements, and the limited adaption to growth in cases of pediatric patients. The ability to replace failing pacemaker tissue with functional pacemaker cells derived from stem cells to generate a biological pacemaker represents an attractive alternative that could overcome these disadvantages. We have previously been successful in generating SAN pacemaker cells from stem cells and have shown that these cells can function as a biological pacemaker in a small animal model. In this study we aim to identify methods to derive AVN pacemaker cells from stem cells and to functionally characterize their pacemaker phenotype. The long-term objective is to use these AVN pacemaker cells in biological pacemaker approaches to provide an improved treatment for patients with AVN pacemaker diseases in Canada and worldwide.
The human heart rate is controlled by two specialized pacemaker tissues in the heart known as the sinoatrial node (SAN) and the atrioventricular node (AVN). The SAN initiates electrical signals that drive the heartbeat while the AVN is responsible for propagating these electric signals from the upper heart chambers to the bottom heart chambers. This activates their contraction and results in the pumping of blood through the body. Failure of either pacemaker tissue leads to a life-threatening irregular or slow heartbeat and needs to be treated by implantation of an electronic pacemaker device. Approximately 21,000 Canadians receive an electronic pacemaker every year. While representing an effective treatment, electronic pacemakers have a number of disadvantages including the risk of pacemaker lead infections, the need for surgical battery replacements, and the limited adaption to growth in cases of pediatric patients. The ability to replace failing pacemaker tissue with functional pacemaker cells derived from stem cells to generate a biological pacemaker represents an attractive alternative that could overcome these disadvantages. We have previously been successful in generating SAN pacemaker cells from stem cells and have shown that these cells can function as a biological pacemaker in a small animal model. In this study we aim to identify methods to derive AVN pacemaker cells from stem cells and to functionally characterize their pacemaker phenotype. The long-term objective is to use these AVN pacemaker cells in biological pacemaker approaches to provide an improved treatment for patients with AVN pacemaker diseases in Canada and worldwide. Chronic back pain associated with intervertebral disc degeneration is a debilitating condition that negatively impacts the quality of life of many Canadians. Current treatment strategies focusing on pain management or surgical intervention have limited efficacy and can lead to numerous complications. Cell-based therapies involving the delivery of pro-regenerative stem cells into the intervertebral disc (IVD) hold promise as a new approach to modify the progression of the disease and promote constructive tissue repair, with the goal of improving function and reducing pain. This research project brings together a multidisciplinary research team to develop novel cell-instructive biomaterials for applications in cell delivery to promote IVD regeneration, as well as in advanced models of IVD disease. These biomaterials platforms will be designed to mimic the composition of the human IVD within a naturally-derived polymer that forms a gel at body temperature. Through detailed cell culture studies, the team will explore the proliferation and differentiation of human nucleus pulposus cells and notochord-like stem cells following encapsulation, and will tune the platform to enhance cell viability and lineage-specific differentiation to augment the regenerative potential of the cells. Future studies will extend the approach to generate tissue-specific disease models to develop a better understanding of the cellular changes that occur in IVD degeneration and the impact of the diseased microenvironment on IVD cells. Our platforms will provide much-needed insight into the effects of the local microenvironment on IVD cell function and phenotype and may be applied in future stem cell-based therapies for the treatment of IVD degeneration.
Chronic back pain associated with intervertebral disc degeneration is a debilitating condition that negatively impacts the quality of life of many Canadians. Current treatment strategies focusing on pain management or surgical intervention have limited efficacy and can lead to numerous complications. Cell-based therapies involving the delivery of pro-regenerative stem cells into the intervertebral disc (IVD) hold promise as a new approach to modify the progression of the disease and promote constructive tissue repair, with the goal of improving function and reducing pain. This research project brings together a multidisciplinary research team to develop novel cell-instructive biomaterials for applications in cell delivery to promote IVD regeneration, as well as in advanced models of IVD disease. These biomaterials platforms will be designed to mimic the composition of the human IVD within a naturally-derived polymer that forms a gel at body temperature. Through detailed cell culture studies, the team will explore the proliferation and differentiation of human nucleus pulposus cells and notochord-like stem cells following encapsulation, and will tune the platform to enhance cell viability and lineage-specific differentiation to augment the regenerative potential of the cells. Future studies will extend the approach to generate tissue-specific disease models to develop a better understanding of the cellular changes that occur in IVD degeneration and the impact of the diseased microenvironment on IVD cells. Our platforms will provide much-needed insight into the effects of the local microenvironment on IVD cell function and phenotype and may be applied in future stem cell-based therapies for the treatment of IVD degeneration. One of the major complications of diabetes is the alteration of wound healing, which can lead to chronic wounds such as diabetic foot ulcers. Diabetes prolongs the duration of normal wound healing by shifting neutrophil infiltration and monocyte/macrophages polarization towards pro-inflammatory subtypes. Mesenchymal stromal cells (MSCs) are a potential therapeutic option for diabetic wound complications. Recently, MSCs have been utilized for their anti-inflammatory functionality in Crohn’s disease, graft vs. host disease and critical limb ischemia. The Viswanathan lab has developed a novel protocol to enhance MSCs by growing them in scalable 3D suspensions in serum-free medium and proprietary cytokines or small molecules. The 3D configuration alters cell adhesion markers and results in more potent anti-inflammatory and immune modulatory MSCs without licensing or genetic modification. Importantly, the enhanced anti-inflammatory and immunomodulatory properties are maintained through cryopreservation, making 3D enhanced MSCs attractive as an off-the-shelf, ready-to use therapy for acute applications, such as diabetic ulcers. In this project, we will focus on optimizing cryopreservation parameters for 3D configurations of MSCs. Using design-of-experiment statistical approaches, we will systematically optimize combinations of parameters to cryopreserve 3D configurations of MSCs for immediate post-thaw use in a validated diabetic ulcer model in collaboration with the Rogers lab. These studies will provide proof-of-concept data for clinical trial applications using potent, ready-to use MSC products for diabetic wounds.
One of the major complications of diabetes is the alteration of wound healing, which can lead to chronic wounds such as diabetic foot ulcers. Diabetes prolongs the duration of normal wound healing by shifting neutrophil infiltration and monocyte/macrophages polarization towards pro-inflammatory subtypes. Mesenchymal stromal cells (MSCs) are a potential therapeutic option for diabetic wound complications. Recently, MSCs have been utilized for their anti-inflammatory functionality in Crohn’s disease, graft vs. host disease and critical limb ischemia. The Viswanathan lab has developed a novel protocol to enhance MSCs by growing them in scalable 3D suspensions in serum-free medium and proprietary cytokines or small molecules. The 3D configuration alters cell adhesion markers and results in more potent anti-inflammatory and immune modulatory MSCs without licensing or genetic modification. Importantly, the enhanced anti-inflammatory and immunomodulatory properties are maintained through cryopreservation, making 3D enhanced MSCs attractive as an off-the-shelf, ready-to use therapy for acute applications, such as diabetic ulcers. In this project, we will focus on optimizing cryopreservation parameters for 3D configurations of MSCs. Using design-of-experiment statistical approaches, we will systematically optimize combinations of parameters to cryopreserve 3D configurations of MSCs for immediate post-thaw use in a validated diabetic ulcer model in collaboration with the Rogers lab. These studies will provide proof-of-concept data for clinical trial applications using potent, ready-to use MSC products for diabetic wounds. Micro-encapsulation of stem cells has proven to be a practical methodology for improving cell engraftment and repair of injured heart muscle. However, to date, most microcapsules have been prepared using inert polymeric matrices and/or synthetic polymeric blends that limit its biodegradability and makes clinical translation difficult. In this project, the team will develop and assess the in vivo performance of a next generation of collagen-based microcapsules for progenitor cell therapies specifically designed to improve capsule retention and biodegradability within injured cardiac (heart) tissue. We hypothesize that micro-engineering the compositional structure of capsules, using medical grade collagen will produce materials with enhanced the length of time the cells are retained at the site of injection and biological activity within infarcted myocardium (tissues damaged by a heart attack). Since the new capsules are cleavable ; this provides a means to deliver stem cells selectively in regions where cardiac functional remodelling is taking place. Thus, our novel materials will open new avenues towards the development of bio-responsive materials for boosting functional engraftment of stem-cells within injured cardiac muscle.
Micro-encapsulation of stem cells has proven to be a practical methodology for improving cell engraftment and repair of injured heart muscle. However, to date, most microcapsules have been prepared using inert polymeric matrices and/or synthetic polymeric blends that limit its biodegradability and makes clinical translation difficult. In this project, the team will develop and assess the in vivo performance of a next generation of collagen-based microcapsules for progenitor cell therapies specifically designed to improve capsule retention and biodegradability within injured cardiac (heart) tissue. We hypothesize that micro-engineering the compositional structure of capsules, using medical grade collagen will produce materials with enhanced the length of time the cells are retained at the site of injection and biological activity within infarcted myocardium (tissues damaged by a heart attack). Since the new capsules are cleavable ; this provides a means to deliver stem cells selectively in regions where cardiac functional remodelling is taking place. Thus, our novel materials will open new avenues towards the development of bio-responsive materials for boosting functional engraftment of stem-cells within injured cardiac muscle. There are two forms of diabetes mellitus: type 1 diabetes (T1D), characterized by the destruction of insulin-producing cells (β cells) by the immune system and type 2 diabetes (T2D), a metabolic disorder that is linked to insulin resistance and β cell failure. Currently, T1D is treated by insulin administration using needle injections or pumps. Patients with T2D are treated with medications that increase insulin secretion from the β cells, reduce sugar production by the liver or enhance insulin sensitivity. Despite these treatments, poorly controlled sugar levels in the blood leads to severe complications, including blindness, limb amputations, kidney failure and cardiovascular diseases. According to Statistics Canada, diabetes mellitus is the 6th leading cause of death in Canada. It is clear that in order to improve these figures, new generations of therapies will be required. The recent progress in stem cell research is bringing us closer to developing β cell therapies for T1D. This proposal leverages on Drs. Nostro and Keller’s expertise on the differentiation of insulin-producing pancreatic β cells and hematopoietic cells. Together, we propose to profile various cell types in the developing human pancreas to identify novel regulatory mechanisms that control β cell maturation and use this information to optimize the production of β cells from human pluripotent stem cells (hPSCs), generating a cell type that has potential for transplantation into patients with T1D.
There are two forms of diabetes mellitus: type 1 diabetes (T1D), characterized by the destruction of insulin-producing cells (β cells) by the immune system and type 2 diabetes (T2D), a metabolic disorder that is linked to insulin resistance and β cell failure. Currently, T1D is treated by insulin administration using needle injections or pumps. Patients with T2D are treated with medications that increase insulin secretion from the β cells, reduce sugar production by the liver or enhance insulin sensitivity. Despite these treatments, poorly controlled sugar levels in the blood leads to severe complications, including blindness, limb amputations, kidney failure and cardiovascular diseases. According to Statistics Canada, diabetes mellitus is the 6th leading cause of death in Canada. It is clear that in order to improve these figures, new generations of therapies will be required. The recent progress in stem cell research is bringing us closer to developing β cell therapies for T1D. This proposal leverages on Drs. Nostro and Keller’s expertise on the differentiation of insulin-producing pancreatic β cells and hematopoietic cells. Together, we propose to profile various cell types in the developing human pancreas to identify novel regulatory mechanisms that control β cell maturation and use this information to optimize the production of β cells from human pluripotent stem cells (hPSCs), generating a cell type that has potential for transplantation into patients with T1D.
 The single most important factor that determines survival of a burn patient is wound healing. This team recently isolated cells identified as burn-derived mesenchymal stem cells (BD-MSCs) from discarded burned skin. A skin substitute, Integra®-SC, was engineered by incorporating BD-MSCs into Integra®, a dermal matrix, and found beneficial results in both small and large animal models. In this study the team will conduct a first-in-humans clinical trial using Integra®-SC, developed from a patient’s own surgically removed burned tissue. It’s believed that Integra®-SC will facilitate and improve wound healing, heal faster, and, in the long-term, result in less scar formation. This approach also reduces the burden of surgeons needing to surgically remove a patient’s own good uninjured skin to use as a donor. This study will be conducted in stages followed by interim safety analyses. After safety analyses, the team will evaluate any needs or gaps in order to include recruitment of patients with a larger percent total body surface area burn. Integra®-SC has a broad clinical application and will impacts the larger burn community, patients with traumatic and complex wounds, and the stem cell research community. The clinical applicability of these results is promising and could create a new standard for the way we care for patients in the province of Ontario and world-wide.
The single most important factor that determines survival of a burn patient is wound healing. This team recently isolated cells identified as burn-derived mesenchymal stem cells (BD-MSCs) from discarded burned skin. A skin substitute, Integra®-SC, was engineered by incorporating BD-MSCs into Integra®, a dermal matrix, and found beneficial results in both small and large animal models. In this study the team will conduct a first-in-humans clinical trial using Integra®-SC, developed from a patient’s own surgically removed burned tissue. It’s believed that Integra®-SC will facilitate and improve wound healing, heal faster, and, in the long-term, result in less scar formation. This approach also reduces the burden of surgeons needing to surgically remove a patient’s own good uninjured skin to use as a donor. This study will be conducted in stages followed by interim safety analyses. After safety analyses, the team will evaluate any needs or gaps in order to include recruitment of patients with a larger percent total body surface area burn. Integra®-SC has a broad clinical application and will impacts the larger burn community, patients with traumatic and complex wounds, and the stem cell research community. The clinical applicability of these results is promising and could create a new standard for the way we care for patients in the province of Ontario and world-wide.




 Damage to the white matter of the brain occurs following injury and in disorders like multiple sclerosis. This results in sensory, motor, and cognitive problems. Currently, there are no effective medical therapies to promote brain repair and reduce disability following white matter damage. This project looks at enhancing the genesis of new oligodendrocytes (the cells that make myelin) and in doing so, promote white matter repair. Taking advantage of the fact that our brains contain resident precursor cells that normally make oligodendrocytes throughout life, the team will ask whether endogenous precursors can be pharmacologically activated to promote repair. Excitingly, this team has discovered that a widely-used and safe drug, metformin, will enhance the genesis of oligodendrocytes from neural precursors and that, following pediatric neural injury, this promotes neuroanatomical and functional recovery. This work will now be translated to the clinic. The team is pursuing a pilot clinical trial of metformin for pediatric demyelinating disease using outcome measures recently developed. Positive results from this clinical trial will lead to a dramatic shift in how we treat children/teenagers with white matter injury, and will pave the way for future additional clinical trials in children and adults with white matter damage.
Damage to the white matter of the brain occurs following injury and in disorders like multiple sclerosis. This results in sensory, motor, and cognitive problems. Currently, there are no effective medical therapies to promote brain repair and reduce disability following white matter damage. This project looks at enhancing the genesis of new oligodendrocytes (the cells that make myelin) and in doing so, promote white matter repair. Taking advantage of the fact that our brains contain resident precursor cells that normally make oligodendrocytes throughout life, the team will ask whether endogenous precursors can be pharmacologically activated to promote repair. Excitingly, this team has discovered that a widely-used and safe drug, metformin, will enhance the genesis of oligodendrocytes from neural precursors and that, following pediatric neural injury, this promotes neuroanatomical and functional recovery. This work will now be translated to the clinic. The team is pursuing a pilot clinical trial of metformin for pediatric demyelinating disease using outcome measures recently developed. Positive results from this clinical trial will lead to a dramatic shift in how we treat children/teenagers with white matter injury, and will pave the way for future additional clinical trials in children and adults with white matter damage.
